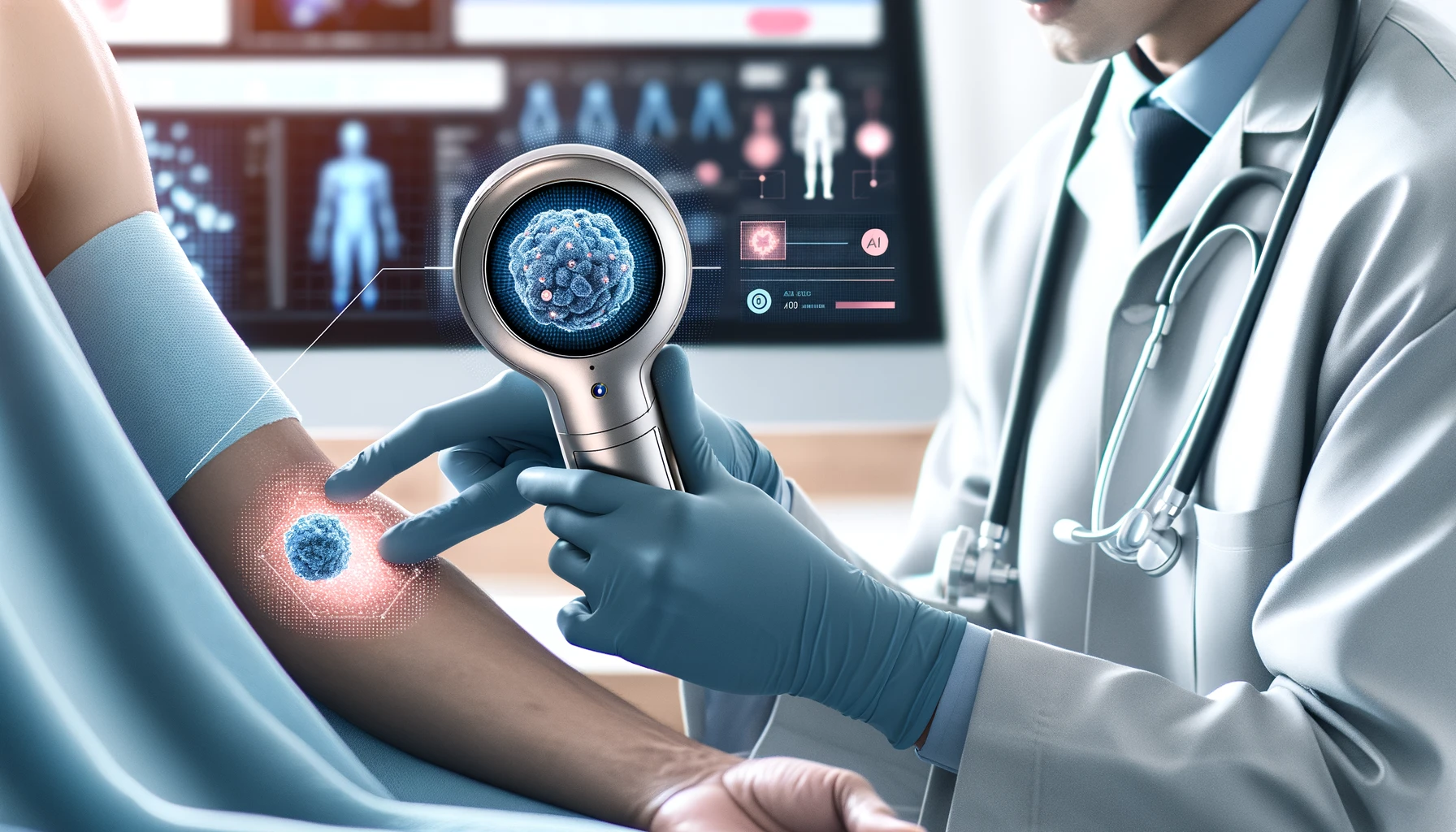
The U.S. Food and Drug Administration (FDA) has recently authorized the marketing of the DermaSensor Inc. DermaSensor device, a potential advancement in the detection and evaluation of skin cancer. This AI-powered hand-held device is designed to assist healthcare providers in evaluating skin lesions suggestive of melanoma, basal cell carcinoma, and squamous cell carcinoma in patients aged 40 and over.
The DermaSensor device is a prescription tool intended to aid non-dermatologist physicians in determining whether a patient should be referred to a dermatologist. It uses a combination of AI and light-based technology to analyze skin lesions that have already been assessed as suspicious for skin cancer. The device shines light on the skin and employs AI algorithms to differentiate between benign and malignant lesions, based on the reflected signals.
While DermaSensor offers a more accurate assessment of skin lesions, it is important to note that it is not intended as a standalone diagnostic tool or a screening device. It should be used in conjunction with a comprehensive clinical assessment, including a visual analysis of the lesion. The device is specifically designed for use on lesions already deemed suspicious for skin cancer and should not be the sole criterion for diagnosing the disease.
Recognizing the need for broader clinical validation, the FDA has mandated additional post-market performance testing of the DermaSensor device. This testing aims to ensure its effectiveness across diverse demographic groups representative of the U.S. population, including those with a relatively low incidence of melanoma, who were underrepresented in the premarket studies.
For healthcare providers, particularly those who are not dermatologists, DermaSensor offers a valuable tool in the early detection of skin cancer, potentially leading to timely and life-saving interventions. For patients, especially those over 40, it provides an additional layer of assessment, enhancing the chances of catching skin cancer in its early stages.
The post “FDA approves AI-powered DermaSensor for enhanced skin cancer evaluation” by Maxwell William was published on 01/19/2024 by readwrite.com